1) Pregnant related pathologies:
ECTOPIC pregnancy
Ectopic pregnancy refers to the implantation of a gestational sac outside of the uterine cavity.
The overall incidence is 1-2% of pregnancies [3].
The classic clinical presentation is with abdominal pain,
amenorrhea and bleeding.
Initial evaluation consists of hormonal assays and pelvic ultrasonography (US).
Symptoms could not be necessarily severe: mild pelvic pain and hemorrhagic spotting in a patient in early pregnancy (5-9 weeks of amenorrhea) [4,5].
Nonetheless,
monitoring of hemodynamic status is crucial,
as hemorrhage can be life threatening: hypovolemic shock and shoulder pain secondary to diaphragmatic irritation are indirect signs of a ruptured ectopic pregnancy.
Any clinical suspicion for a ruptured ectopic pregnancy in a patient in unstable conditions warrants emergent surgical intervention.
Early diagnosis and treatment of ectopic pregnancy are essential in reducing maternal mortality and preserving future fertility.
Ectopic implantation site are shown in Table 3 .
In ectopic pregnancies,
serum hCG levels often rise at a much slower rate: if β-hCG levels increase by less than 50% during a 48-hour period,
there is almost always a nonviable pregnancy associated,
be it intra- or extrauterine [6].
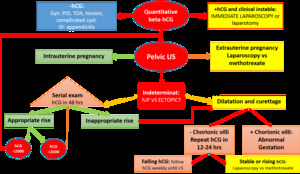
Fig. 1: Ectopic management algorithm adapted from D.E. McWilliams, DOa, Micah J. Hill, Charles S. Dietrich III, Gynecologic Emergencies. Surg Clin N Am 88 (2008) 265–283
85% percent of viable intrauterine pregnancies reflect an increase in β-hCG levels of 66% or more during a 48-hour period [7].
However,
up to 21% of ectopic pregnancies demonstrate a β-hCG doubling time identical to that of intrauterine pregnancies [8].
Arriving at a β-hCG plateau early in the pregnancy is highly suggestive of an ectopic pregnancy.
It is useful to know a quantitative beta HCG prior to scanning as this will guide what you expect to see.
Transvaginal US should be able to demonstrate a gestational sac when β-hCG levels are greater than 2000 mIU/mL,
which is the discriminatory level of β-hCG [9].
However,
some institutions may use a higher threshold.
Transabdominal US can demonstrate an intrauterine pregnancy when hCG levels reach 6500 mIU/mL.
The ultrasound examamination should be performed both trans abdominally and transvaginally.
The trans abdominal component provides a wider overview of the abdomen,
whereas a transvaginal scan is important for diagnostic sensitivity.
Positive sonographic findings include:
Uterus:
- empty uterine cavity or no evidence of intrauterine pregnancy;
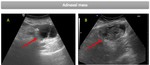
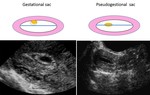
- pseudogestational sac or decidual cyst: may be seen in 10-20% of ectopic pregnancies,
remember “double decidual sac sign” (Fig. 2 );
-thick echogenic endometrium;
Tube and ovary
- simple adnexal cyst: 10% chance of an ectopic
- complex extra-adnexal cyst/mass: 95% chance of a tubal ectopic (if no IUP)
- an intra-adnexal cyst/mass is more likely to be a corpus luteum
- "ring of fire" can be seen on color Doppler in a tubal ectopic pregnancy,
but can also be seen in a corpus luteum
- absence of colour Doppler flow does not exclude an ectopic pregnancy.
- live extrauterine pregnancy (i.e.
extra-uterine fetal cardiac activity): 100% specific.
A pseudogestational sac is the idea that a small amount of intrauterine fluid in the setting of a positive pregnancy test and abdominal pain could be erroneously interpreted as a true gestational sac in ectopic pregnancy Fig. 2 .
The sign was originally reported before the use of endovaginal ultrasound imaging and caution should be exercised if considering diagnosing a pseudogestational sac with modern ultrasound equipment.
In a woman with a postive beta-hCG,
any intrauterine sac-like fluid collection seen on ultrasound is highly likely to be a gestational sac.
Although ectopic pregnancy is usually diagnosed on the basis of a combination of clinical,
laboratory,
and ultrasonographic findings,
it is occasionally and initially identified at MR imaging.
Ectopic pregnancy is increasingly depicted with MR imaging,
usually unexpectedly in the clinical setting of suspected appendicitis or,
less commonly,
in the workup of a known ectopic pregnancy.
MR imaging can be a useful problem-solving adjunct to US because MR imaging provides images with high soft-tissue contrast,
which sometimes help to localize the ectopic gestational sac when it is not identified with US.
Some example of varied appearances of ectopic pregnancy are:
· Lack of Intrauterine Pregnancy;
· Hemoperitoneum: for this reason inclusion of a T1-weighted pulse sequence is helpful in identifying blood products and is recommended as part of any imaging protocol especially techniques that take advantage of T2 shortening from magnetic susceptibility effects (T2*- and susceptibility-weighted MR imaging);
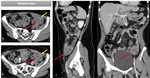
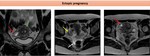
· Tubal Mass: the predominant initial finding is a heterogeneous predominantly high-signal-intensity mass on T2-weighted MR images.
The mass may appear as a thick walled ring with small cystic spaces ( Fig. 3).
· Hematosalpinx: The presence of hematosalpinx in a woman with positive results of a pregnancy test and with no intrauterine gestational sac is highly concerning for ectopic pregnancy,
even in the absence of a clearly identifiable extrauterine gestational sac.
Important to note that terms interstitial pregnancy and cornual pregnancy are not synonymous of angular pregnancy [10,
11] and the correct diagnosis in essential for better managment of this patient.
The term cornual pregnancy is often reserved to implantation in one horn of a bicornuate or septate uterus.
Interstitial pregnancy refers to implantation in the most proximal segment of the fallopian tube where it traverses the uterine musculature meanwhilr angular pregnancy is defined as implantation in one of the lateral angles of the uterus,
medial to the uterotubal junction and the round ligament of the uterus.
MRI can have a role in the differential diagnosis due to its high spatial and panoramic resolution.
The differential diagnosis is important because:
· angular pregnancies can sometimes be carried to term; but i is associated with high rates of spontaneous abortion,
uterine rupture,
and placenta accreta,
with rupture occurring in as many as 23.5% of all angular pregnancies ([12]. Due to intrauterine location patients present symptoms later than patients affected by other types of ectopic pregnancies.
· Interstitial pregnancy due to its proximity to the uterine artery,
it is associated with marked hemoperitoneum (maternal mortality rate for interstitial pregnancy is high 2.5%).
Moreover Clinicians have myometrium surrounding the interstitial segment of the tube allows a greater distention before rupture for this reason interstitial ectopic pregnancies progress further into gestation before rupture,
perhaps to 12–16 weeks.
Fig. 3
Non pregnant related pathologies
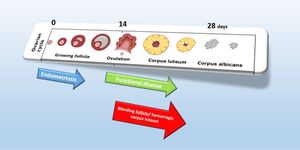
Fig. 14: Cycle-phase related pathologies
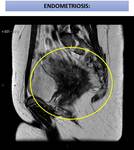
a. Menstrual related: Endometriosis
Endometriosis,
which is defined as the presence of ectopic endometrial glands and stroma outside the uterus,
is a common cause of pelvic pain and infertility,
affecting as many as 10% of premenopausal women [13].
Onset of pain usually precedes flow by a few days and begins to resolve 1-2 days into the menses.
Pelvic pain,
including dyspareunia,
dysmenorrhea could be also not cyclic.
Acute exacerbations are believed to be caused by chemical peritonitis due to leakage of old blood from an endometriotic cyst.
Unusual symptoms can be:
o gastrointestinal involvement: catamenial diarrhoea,
rectal bleeding and constipation,
o urinary system involvement: urgency,
frequency,
haematuria and hydroureteronephrosis,
o thoracic involvement: pleuritic chest pain,
pneumothorax,
pleural effusions or cyclic hemoptysis.
o asymptomatic: especially if the disease is isolated to the peritoneum.
Ultrasound detects only 11% of peritoneal implants,
and although it is unable to detect adhesions,
it is able to dynamically assess mobility and fixation.
On the other hand,
Ultrasound is a good technique to detect endometriomas.
US features:
· homogenous,
focal lesions with low-level echoes,
reminiscent of the testicle;
· unilocular (typically),
multilocular,
containing thin or thick septations;
· single or multiple
· may contain mural nodules (a finding also found in ovarian neoplasms),
if these mural nodules are hyperechoic,
these have a high predictive value for endometrioma over non-endometrial lesions
· colour Doppler: no internal vascularity
· as opposed to many other ovarian cysts,
endometriomas do not resolve.
Despite repeated hemorrhage,
findings of acute hemorrhage are uncommon in endometriomas (<10%),
such as layering blood products or retractile thrombus.
MRI has a greater specificity for the diagnosis not only of endometriomas and allows also a better comprehensive evaluation of disease extension than the other non-invasive imaging techniques.
has a role to play in the evaluation of adnexal masses,
as well as assessing for response to medical therapy.
Typically the lesions that can be detected with MRI are those that contain blood products.
Figure describes MR imaging appearances that may be helpful for differentiating endometriomas and deep pelvic endometriosis from other causes of pelvic pain and infertility in women.
- endometriomas,
and haemorrhagic “powder burn”, Fig. 4,
Fig. 5
- adhesions,
fibrosis and uterosacral involvement Fig. 6 Fig. 7
- vaginal involvement /pouch of Douglas / rectovaginal septum Fig. 8
- gastrointestinal tract Fig. 9
- cutaneous tissues/nodules Fig. 10,
Fig. 11
- malignant transformation ( Fig. 12) high specific finding is development of enhancing mural nodules
Women with endometriosis are at risk for developing both clear cell and endometrioid subtypes of epithelial ovarian cancer [14].
An estimated 2.5% of women with endometriosis develop ovarian cancer.
Differential considerations on MR imaging for endometriomas include:
- dermoid cyst: endometriomas have homogeneous high signal intensity on T1 which does not suppress on T1FS, unlike a dermoid which has signal drop out on fat suppression images and chemical shift artefact
- mucinous lesions: e.g.
ovarian mucinous tumors; increased signal on T1 but less intense than fat or blood.
Fig. 13
- hemorragic ovarian cysts: endometriomas rarely present with acute symptoms and do not resolve over time.
An important MRI sign is Shading sign: it is seen in an endometrioma.
It may also be seen with some endometrioid tumours (e.g endometrioid carcinoma of the ovary).
It helps to distinguish endometriomas from other blood containing lesions (e.g.
hemorrhagic corpus luteum cysts) [15]
The degree of shading can vary from faint to complete signal loss.
When noted with endometriosis,
lesions demonstrating a shading sign may be less likely to respond to medical treatment [16].

Table 4: Differential diagnosis between Endometrioma and hemorrhagic cyst-.
a. Ovulation related:
Ovary imaging aspect change physiologically with cycle phase due to endocrine stimulation,
Fig. 15 .
Rupture of an ovarian follicle (14 day of cycle) is a physiologic event which can be painful (mittelschmerz,
‘middle pain’)
A hemorrhagic or a ruptured ovarian cyst is the most common cause of acute pelvic pain in an afebrile,
premenopausal woman presenting to the emergency room.
They can occur during pregnancy.
Rupture of an ovarian cyst (>3 cm) may cause more dramatic clinical symptoms.
The pain due to the ruptured ovarian cyst may come from [18]:
· stretching the capsule of the ovary,
· torqueing of the ovarian pedicle,
· Leakage of cyst contents (serous fluid/blood) which can cause peritoneal irritation.
Bleeding follicle
Pelvic pain during the ovulatory cycle may be observed due to a small amount of blood which drains from the ruptured ovarian follicle to the peritoneal cavity during ovulation.
This pain is mild-to-moderate and limited,
and hemoperitoneum is observed with normal hemostatic parameters.
Thus,
there is generally no need for surgical intervention in these setting.
Hemorrhagic ovarian cysts (HOCs) usually result from hemorrhage into a corpus luteum or other functional cyst.
Radiographic features are variable and depends on the age of the hemorrhage.
They typically resolve within eight weeks. Patients may present with sudden-onset pelvic pain,
pelvic mass,
or they may be asymptomatic and the HOC is an incidental finding .
HOCs can have a variety of appearances depending on the stage of evolution of the blood products and clot Fig. 16 :
- lace-like reticular echoes or an intracystic solid clot (fluid-fluid level is possible);
- thin wall;
- clot may adhere to the cyst wall mimicking a nodule,
but no blood flow on Doppler imaging is present;
- retracting clot may have sharp or concave borders,
- no any internal blood flow;
- circumferential blood flow in the cyst wall is typical.
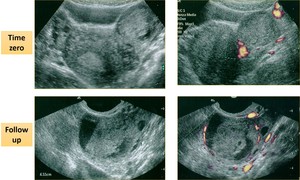
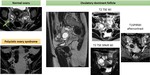
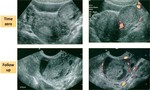
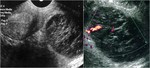
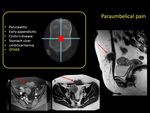
Fig. 16: 40 years old woman presented to the Emergency room fo lower quadrants pain:
US investigation showed solid ovarian lesion (4 cm of diameter) with there were not any internal blood flow.
After 3 weeks, a second US examination was performed and it showed a pseudo-solid lesion without internal blood flow and circumferential blood flow in the cyst wall.
Findings were typical for corpus luteum bleeding.
References: Radiology Department, ASL2 Savona.
CT investigation can also visualised ruptured of corpus luteal cyst as a low-attenuating cyst (25 HU),
<3 cm in diameter with thick slightly irregular iso- or hyperattenuating walls.
Free fluid is often associated,
Fig. 17 .
On MRI investigation,
hemorrhagic adnexal cyst (most commonly,
a corpus luteum cyst) may be bright on T1-weighted images,
they are usually solitary and thin walled and are brighter on T2-weighted images than are endometriomas.
Marked loss of signal intensity on T2-weighted images is not usually seen with hemorrhagic cysts,
since they do not repeatedly bleed.
Without recurrent hemorrhage and concentration of contents,
viscosity of the cyst remains lower,
and shading is unlikely to be present.
A cystic structure that does not convincingly satisfy the criteria for a benign cyst cannot be considered a cyst and should be evaluated with a short interval follow-up US or MR.
Especially,
a ruptured hemorragic cyst in a peri-menopausal woman should be viewed more suspiciously and followed up appropriately.
A hemorrhagic cyst or ruptured cyst in a postmenopausal woman deserves surgical evaluation.
For these reasons,
hemorragic cyst have a different follow up schedule than simple cysts (see below),
with ultrasound or MRI in 6-12 weeks if:
· the cyst is > 5 cm in diameter if the patient is pre-menopausal or
· any size of a hemorrhagic cyst if the patient is peri-menopausal.
In the postmenopausal patient,
surgical evaluation is warranted.
Treated conservatively in a premenopausal woman unless evidence of hemorrhagic shock (tachycardia and postural drop in blood pressure).
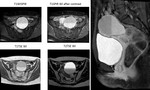
A CURIOUSE CASE OF OVULATION RELATED RIGHT LOWER PAIN is proposed Fig. 18 .
c. Non menstrual related
i. PID (Pelvic inflammatory disease)
Pelvic inflammatory disease (PID) is described as a spread of inflammation from the endometrial cavity and fallopian tubes into the pelvis.
It is a comprehensive term,
which encompasses endometritis,
salpingitis and tubo-ovarian abscesses (TOA).
25% of visits to the emergency departments with gynecological pain are due to PID and diagnosis relies often on clinical data.

Table 5: PID DIAGNOSTIC CRITERIA
Common presentations include:
· acute pelvic pain (of variable intensity),
· cervical motion tenderness,
· vaginal discharge,
· fever and leukocytosis.
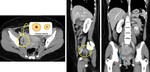
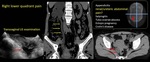
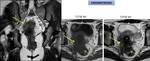
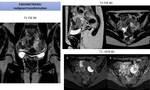
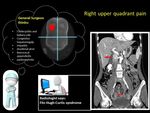
· right upper quadrant pain from the perihepatitis in Fitz-Hugh-Curtis syndrome, Fig. 21 Fig. 22 Fig. 23.
Imaging features are often nonspecific. In the early phase of infection,
CT and US investigations are normal.
If imaged very late,
there may be an adnexal mass-like region with surrounding inflammatory change,
and the fallopian tube and ovary may not be able to be distinguished.
As the disease advances,
transabdominal ultrasound can demonstrate uterine enlargement and thickening of the endometrium.
Some US signs associated with tubal-inflammation include,
thickened/dilated fallopian tubes with:
· incomplete septa in the tube
· increased vascularity around the tube
· echogenic fluid in the tube,
pyosalpinx.
Ultrasound can also show the loss of tissue plains and an ill-defined uterus.
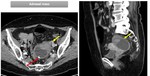
CT and pelvic MRI shows a diffusely-enhancing ill-defined pelvic mass containing fluid,
which may be difficult to differentiate from malignancy,
figure.
The wall and the surrounding tissues may enhance,
Fig. 22 .
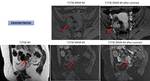
It is very important to recognized complications of PID:
· pyosalpinx formation,
Fig. 19
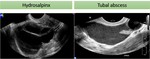
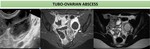
· tubo-ovarian abscesses (TOA) formation,
Fig. 20
· adhesion formation with resultant bowel obstruction
· Fitz-Hugh-Curtis syndrome [17],
Fig. 23.
In the absence of complications,
pelvic inflammatory disease is often treated conservatively with antibiotics.
Hydrosalpinx or pyosalpinx is a common complication of salpingitis.
Typical Ultrasound features of pyosalpinx are dilated fallopian tubes containing heterogenous fluid with echogenic debris.
On MRI,
a pyosalpinx can be visualized as a dilated,
fluid-filled,
tortuous C or S-shaped structure.
Thick-walled fluid-filled abscesses and pyosalpinx may have heterogeneous signal intensity on both T1 and T2 weighting due to mixtures of pus,
hemorrhage and debris.
Pyosalpinx can develop into tubo-ovarian abscesses: the thick-walled mass typically demonstrates marked enhancement following intra-venous gadolinium administration.
Ovarian cancer should always be considered when investigating a possible tubo-ovarian mass.
Imaging features of abscess and ovarian malignancy can be similar: in this setting is the clinical presentation to establish the diagnosis and follow-up imaging following treatment confirms decreasing size of adnexal masses [19,20].
i. Ovarian torsion
Adnexal torsion is the rotation of at least one turn of the ovaries,
adnexa or the fallopian tube around the line of the tubo-ovarian ligament and the infundibulo-pelvic ligament [ 21 ].
It is a real emergency: Urgent surgery is required to prevent ovarian necrosis.
Most patients present with severe nonspecific lower abdominal and pelvic pain,
nausea,
and vomiting.
There is adnexal tenderness. A raised white cell count is common.
Presentation of torsion can be either intermittent or sustained.
Ovarian torsion has a bimodal age distribution occurring mainly in young women (15-30 years) and post-menopausal women.
Two main reasons of torsion are [ 21,23]:
· hypermobility of the ovary: <50% (especially in young women);
· adnexal mass: ~50-80% (dermoid cysts or paraovarian cyst),
masses between 5-10 cm are at most risk.
Approximately 20% of the cases occur during pregnancy.
Most ovaries are not salvageable in which case a salpingo-oophorectomy is required.
If not removed the necrotic ovary can become infected and cause abscess or peritonitis.
Spontaneous de-torsion has also been reported.
Ultrasound,
which is frequently used as the initial imaging investigation,
may detect an adnexal mass,
often in a relatively midline position.
However,
between 9% and 26% of adnexal torsions occurs in normal-sized ovaries and have normal appearances on ultrasound.
Doppler sonography can help to diagnose adnexal torsion (it is specific but not enough sensible): it shows absence of blood flow into the torted ovary.
Important sonographic features include:
· enlarged hypo or hyperechoic ovary (>4 cm)
· peripherally displaced follicles with hyperechoic central stroma
· midline ovary
· free pelvic fluid: may be seen in >80% of cases
· an underlying ovarian lesion may be seen (possible lead point for torsion)
· a long-standing infarcted ovary may have a more complex appearance with cystic or hemorrhagic degeneration
A characteristic “whirlpool sign” may be seen on Color Doppler examination where a corkscrew appearance of a twisted vascular pedicle is apparent.
Remember False-negative reason of Arterial blood flow visualization in adnexal torsion:
-double vascularization of ovary both from ovarian and uterine arteries: only one of the two adnexal arteries (uterine and ovarian) may be torted,
-torsion may be intermittent.
-venous thrombosis secondary to torsion may lead to ovarian necrosis before arterial thrombosis occurs.
CT and MRI may demonstrate a twisted vascular pedicle and thickened fallopian tube in sub-acute torsion,
and identify an underlying mass.
Features of torsion using these modalities include eccentric or concentric wall thickening of the torted adnexal mass,
fallopian tube thickening,
uterine deviation to the effected side and free fluid collection.
HU >50 on non-contrast CT suggests hemorrhagic necrosis and lack of enhancement may be seen.
On MRI,
T1 and fat-suppressed T1-weighted sequence to detect hemorrhage are always essential when using MRI to evaluate an indeterminate adnexal lesion following US.
Hemoperitoneum and hemorrhage with ovarian masses can be demonstrated in other acute emergencies such as a hemorrhagic cysts and rupture.
Other MRI features that can suggest hemorrhagic infarction due to torsion include a thickened cyst wall and low enhancement in the solid component of the twisted ovarian mass.
The ovary may be grossly enlarged due to edema.
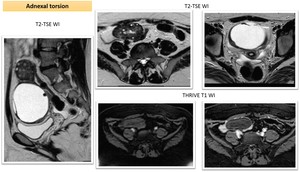
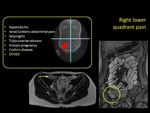
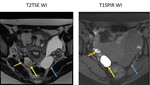
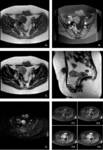
Fig. 24: Young woman with enlarged right torsed ovary. Sagittal and axial MRI T2TSE imaging showed enlarged ovary (4,5 cm) with prevalent hyperintense peripheral follicles , edema of central stroma and a large exophytic cystic mass.
Hemoperitoneum was also present.
THRIVE T1WI before and after contrast administration were performed: no intralesional hemorrhage was detected and no contrast enhancement of ovary was present.
i. Ovarian adnexal mass
Ovarian cancer remains one of the leading causes of cancer-related deaths among women and it is considered as a “silent killer”.
Women with ovarian carcinomas,
often present with vague pelvic and abdominal signs and symptoms,
such as bloating,
early satiety,
abdominal pain and distention,
constipation,
and changes in urinary habits.
Acutely,
patients may present with pleural effusions,
ascites,
small bowel obstructions,
or deep vein thrombosis [25].
Otherwise,
in very rare case,
it can have an acute presentation because of:
o Rupture,
o internal bleeding,
Fig. 25 Fig. 26
o Ovarian torsion,
Fig. 27 .
Some example were show in figure to highlight the importance of this pathology and to not forget it as a possible cause of abdominal pain,
especially in peri-menopausal women.
Also in the clinical setting of Emergency Department,
radiologists have to know suspicious features of cystic masses (Table 9) and correct guideline for the proper management of simple ovarian cysts with no suspicious features on ultrasound which often are only an incidental findings,
table below [24].
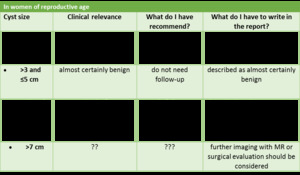
Table 7: table
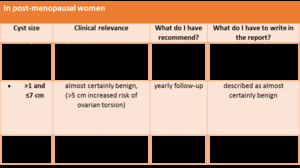
Table 8
As a pratical rule,
the likelihood of malignancy increases with increasing solid-tissue elements and thicker septae. Because of many variations, it may be best to refer to subarticles for specific imaging features [26].

Table 9
And what about CA 125? Can it help us in differential diagnosis?
CA 125 level of 35 units was found to be a useful cut-off point:
< 35 units: 99% of healthy women having values less than 35.
>35 units: suspicious.
Women with ovarian cancer often have levels measured in hundreds and even thousands of units.
Notable exceptions: normal values in patient affected by Stage I ovarian cancer and with certain tumor cell type as mucinous ovarian cancers
Remember that also non-cancerous conditions can elevate CA125 value,
- inflammatory conditions of the abdomen (diverticulitis,
peritonitis,
pelvic inflammatory disease,
inflammatory bowel disease tuberculosis and pancreatitis),
- liver disease,
- recent surgery,
- benign gynecologic conditions such as fibroids,
endometriosis,
ectopic pregnancy,
or a ruptured cyst.
Infact,
CA 125 is also used to monitor the effects of treatment for endometriosis.
These other diagnoses must be considered in the interpretation of an elevated
In conclusion,
CA 125 test is helpful,
but it is better to focus on the trendof the values rather than any individual value.

Table 6


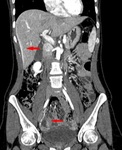
![Fig. 22: PID: Contrast-enhanced CT scan (portal phase) shows uterus (blue arrow), ovaries (red arrows) and the dilated right fallopian tube with thick walls and lumen filled with fluid (green arrow).[17]](https://epos.myesr.org/posterimage/esr/ecr2017/136608/media/681069?maxheight=150&maxwidth=150)